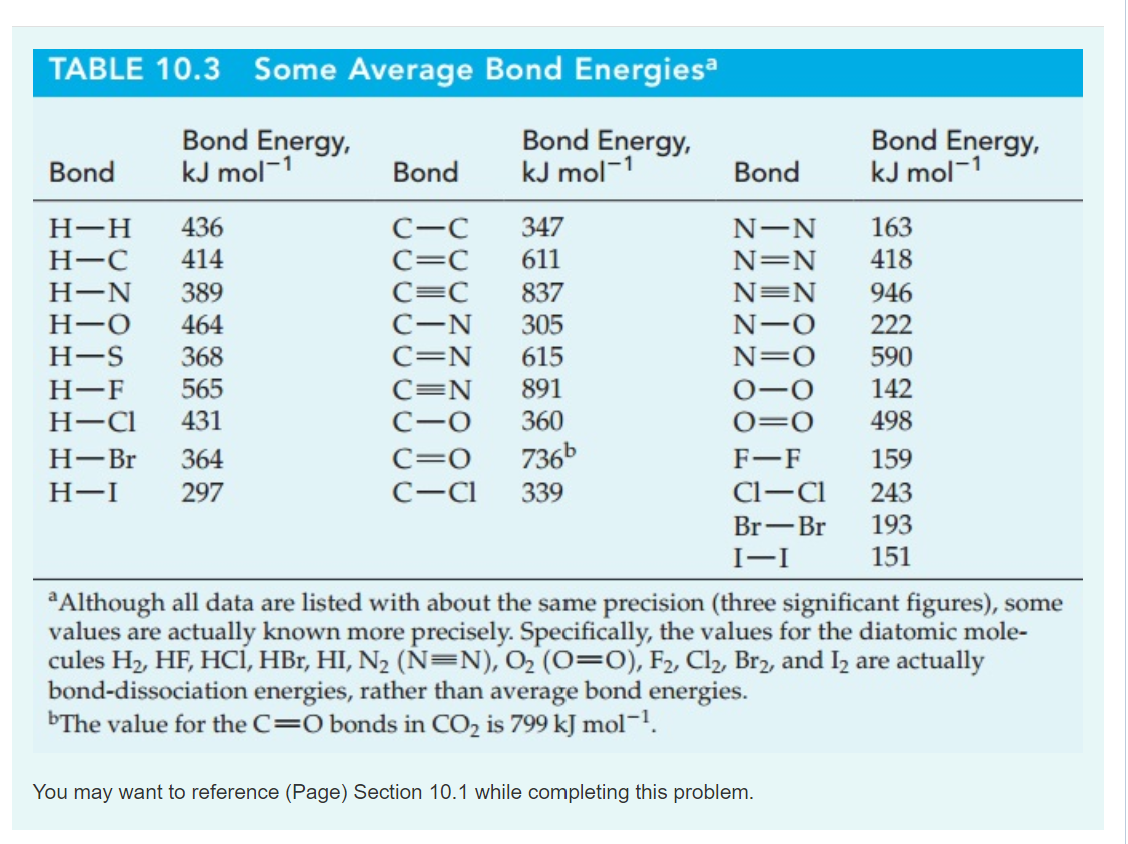
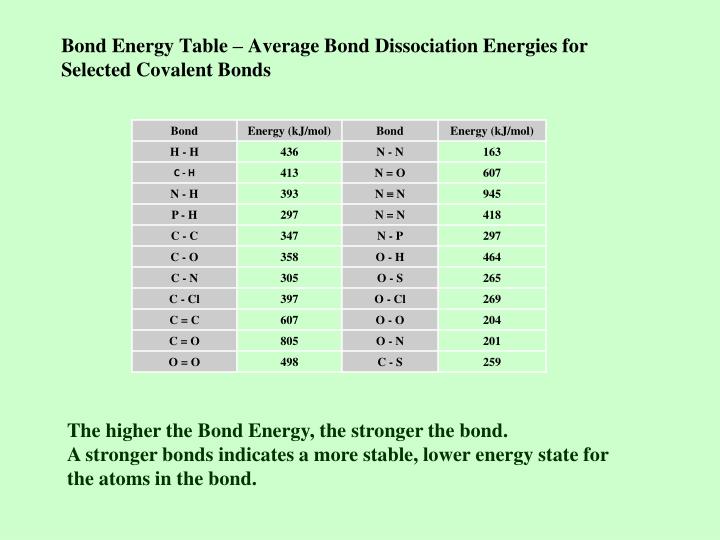
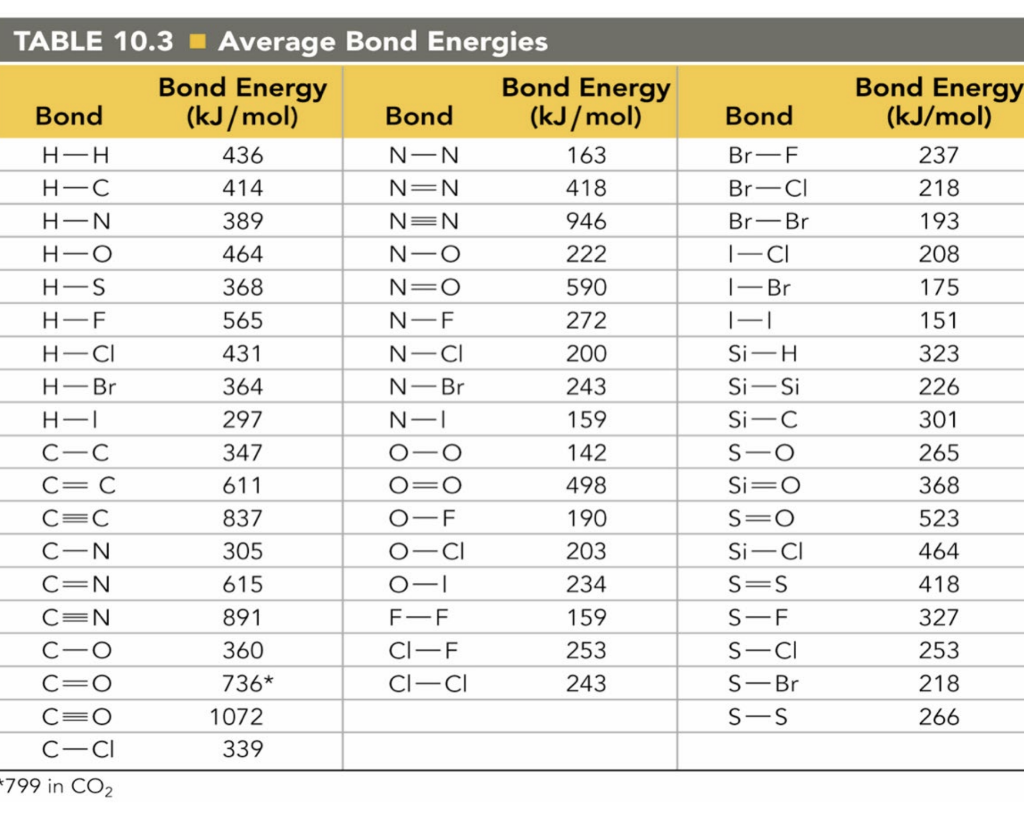
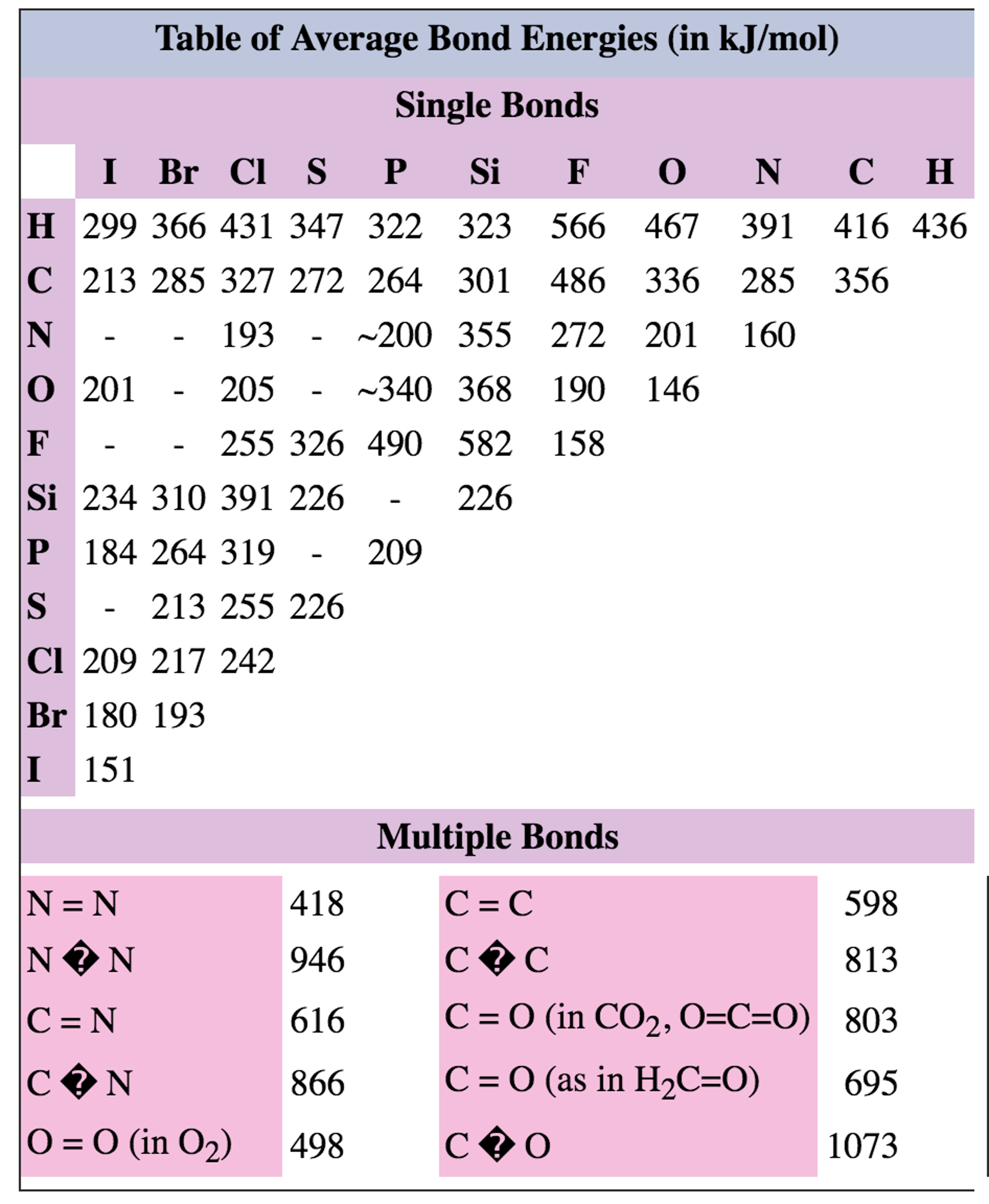
Bond Energies Chart
Bond Energies Chart - Web atoms are held together by a certain amount of energy called bond energy. That is, hf 298 298 298 298 hf (a). Single bonds have a bond order of one, and multiple bonds with bond orders of two (a double bond) and three (a triple bond). Explain its relationship to bond length or bond energy. Mean bond enthalpies are sometimes referred to as bond enthalpy terms. Chemical processes are labeled as exothermic or endothermic based on whether they give off or absorb energy, respectively. Bond energies are also called bond enthalpies, and in the past have been known as bond strengths. Recognize covalent substances and characterize ionic character as difference in electronegativity. Bond strength qualities and periodic trends. Web properties of atoms, radicals, and bonds 4.41 table 4.11 bond dissociation energies the bond dissociation energy (enthalpy change) for a bond a 9b which is broken through the reaction ab : Single bonds have a bond order of one, and multiple bonds with bond orders of two (a double bond) and three (a triple bond). Various qualities determine a bond’s dissociation and formation energies. This page introduces bond energies and looks at how they can be used to estimate the enthalpy change for some simple reactions. You can take all these terms as meaning the same thing. Web bond s— s— f cl energy 218 212 215 bond energy 432 427 363 347 305 358 453 339 276 216 614 (799 in c02) single bonds h— h— h— c— c— c— c— c— c c— c— c c— h c si n o s multiple bonds c n o. We often use a more condensed form of bond energy tables as shown below. Bond order is the number of electron pairs that hold two atoms together. Chemical processes are labeled as exothermic or endothermic based on whether they give off or absorb energy, respectively. Recognize covalent substances and characterize ionic character as difference in electronegativity. The following tables list experimental bond dissociation enthalpies of common bonds at 298 k. This page introduces bond energies and looks at how they can be used to estimate the enthalpy change for some simple reactions. Web the average bond energy is therefore +1662/4 kj, which is +415.5 kj per mole of bonds. Evaluate enthalpies of reactions using bond energies. The exact bond enthalpy of a particular chemical bond depends upon the molecular environment. Web bond s— s— f cl energy 218 212 215 bond energy 432 427 363 347 305 358 453 339 276 216 614 (799 in c02) single bonds h— h— h— c— c— c— c— c— c c— c— c c— h c si n o s multiple bonds c n o. Evaluate enthalpies of reactions using bond energies. Web. Calculate the heat of combustion, for c 2 h 6 using the bond dissociation energies in the table. The exact bond enthalpy of a particular chemical bond depends upon the molecular environment in which the bond exists. (assume complete combustion) check solutions/answers to exercises. This page introduces bond energies and looks at how they can be used to estimate the. The following tables list experimental bond dissociation enthalpies of common bonds at 298 k. Web bond s— s— f cl energy 218 212 215 bond energy 432 427 363 347 305 358 453 339 276 216 614 (799 in c02) single bonds h— h— h— c— c— c— c— c— c c— c— c c— h c si n o. In proposing his theory that octets can be completed by two atoms sharing electron pairs, lewis provided scientists with the first description of covalent bonding. Bond strength qualities and periodic trends. Chemical processes are labeled as exothermic or endothermic based on whether they give off or absorb energy, respectively. Single bonds have a bond order of one, and multiple bonds. (assume complete combustion) check solutions/answers to exercises. You can take all these terms as meaning the same thing. Web this equation can solve for any of these three values and is a key formula in thermodyanamics, physical chemistry, and more. So, ‘’the bond energy is the average amount of energy required to break all bonds of a particular type in. Bond enthalpies are essentially a measure of how strong a covalent bond is. The si units used to describe bond energy is kilojoules per mole of bonds (kj/mol). Web the average bond energy is therefore +1662/4 kj, which is +415.5 kj per mole of bonds. (assume complete combustion) check solutions/answers to exercises. The same amount of energy is absorbed when. Most commonly, a bond’s strength depends on: This is typically the types of tables that are put on exams to save space. That is, hf 298 298 298 298 hf (a). This page introduces bond energies and looks at how they can be used to estimate the enthalpy change for some simple reactions. Bond energies and bond lengths. That means that many bond enthalpies are actually quoted as mean (or average) bond enthalpies, although it might not actually say so. The rest are average bond energies. Most commonly, a bond’s strength depends on: Mean bond enthalpies are sometimes referred to as bond enthalpy terms. The following tables list experimental bond dissociation enthalpies of common bonds at 298 k. Web this page has tables of standard bond energies and bond dissociation energies. You can take all these terms as meaning the same thing. We can calculate a more general bond energy by finding the average of the bond energies of a specific bond in different molecules to get the average bond energy. The following tables list experimental bond dissociation. Bond energies and bond lengths. The exact bond enthalpy of a particular chemical bond depends upon the molecular environment in which the bond exists. The following tables list experimental bond dissociation enthalpies of common bonds at 298 k. You can take all these terms as meaning the same thing. Web properties of atoms, radicals, and bonds 4.41 table 4.11 bond dissociation energies the bond dissociation energy (enthalpy change) for a bond a 9b which is broken through the reaction ab : The si units used to describe bond energy is kilojoules per mole of bonds (kj/mol). Use the bond energies from the table to calculate an approximate δh 0rxn for the following equation: We often use a more condensed form of bond energy tables as shown below. Web this page has tables of standard bond energies and bond dissociation energies. Evaluate enthalpies of reactions using bond energies. Web the bond energy is a measure of the amount of energy needed to break apart one mole of covalently bonded gases. This page introduces bond energies and looks at how they can be used to estimate the enthalpy change for some simple reactions. (assume complete combustion) check solutions/answers to exercises. Web average bond energies *bond dissociation energies for diatomic molecules can be directly measured. * average bond dissociation enthalpies in kcal per mole (there can be considerable variability in some of these values.) Bond energies are also called bond enthalpies, and in the past have been known as bond strengths.Bond Energies Chart
Bond Length and Bond Strength Pathways to Chemistry
The Heat of Reaction from Bond Dissociation Energies Chemistry Steps
Average Bond Energies Chart
Solved Bond TABLE 10.3 Average Bond Energies Bond Energy
Bond Energies Chart
Bond Length and Bond Energy
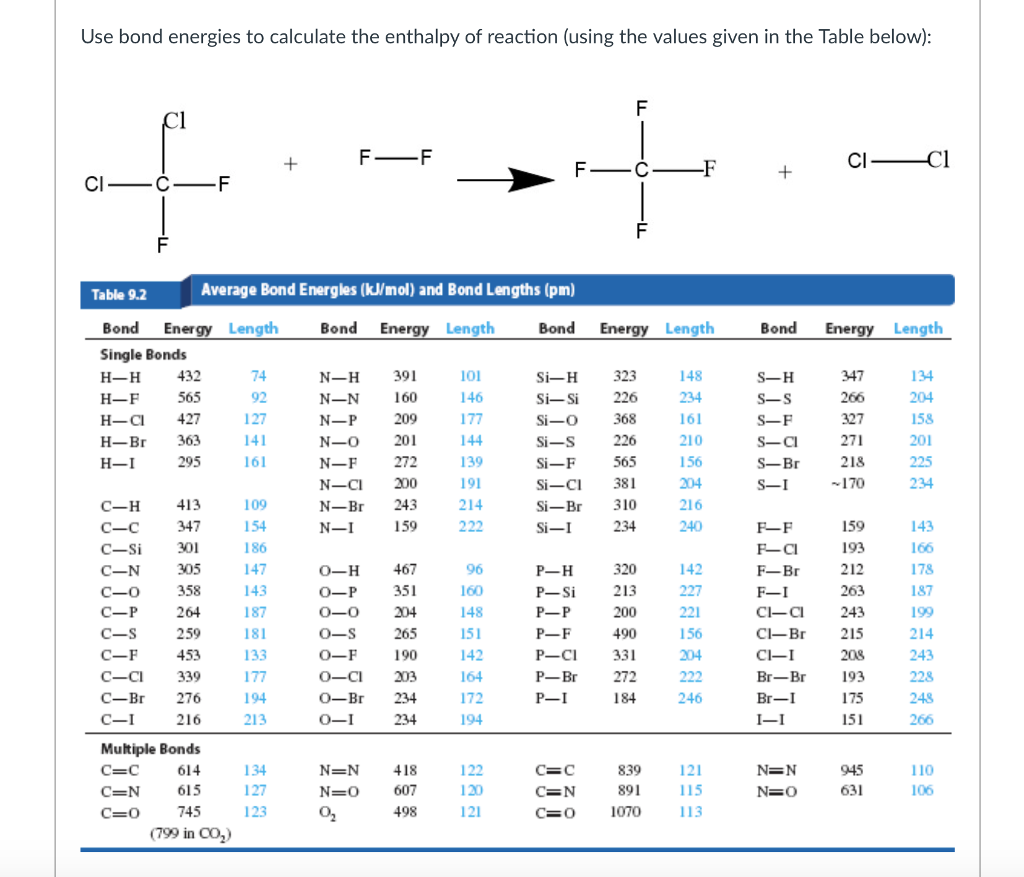
[Solved] Using the appropriate bond energies, calculate the heat of
Table Of Bond Energies Pathways To Chemistry vrogue.co
Bond Energy Profile Chart
Cottrell, The Strengths Of Chemical Bonds, 2Nd Ed., Butterworths, London, 1958;
Web From This Graph, We Can Determine The Equilibrium Bond Length (The Internuclear Distance At The Potential Energy Minimum) And The Bond Energy (The Energy Required To Separate The Two Atoms).
This Is Typically The Types Of Tables That Are Put On Exams To Save Space.
Define Bond Length And Bond Energy And Note The Relationship Between The Two.
Related Post:
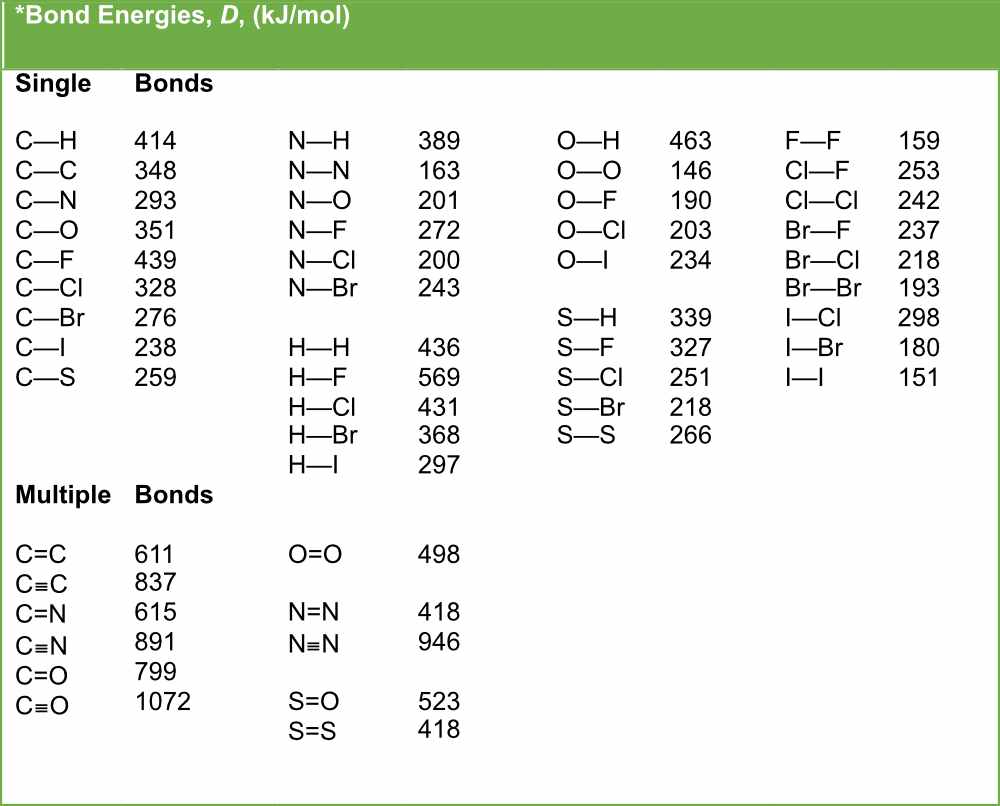


.PNG)